In chemistry, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole moment, with a negatively charged end and a positively charged end. The polarity condition of the electrodes is of two types, (1) straight polarity and (2) reverse polarity. Polarity refers to the electrical conditions determining the direction of the current flow relative to the electrode. This occurs because of a difference in electronegativity of the two atoms that share the electrons.ġ.7 Polarity. Likewise molecules in which there is an accumulation of electron density at one end of the molecule, giving that end a partial negative charge and the other a partial positive charge, are called polar molecules. Nonpolar molecules occur when electrons are shared equal between atoms of a diatomic molecule or when polar bonds in a larger molecule cancel each other out. Polar molecules occur when there is an electronegativity difference between the bonded atoms. While the overall charge of the molecule is neutral, the orientation of the two positively charged hydrogens (+1 each) at one end and the negatively charged oxygen (-2) at the other end give it two poles. Although all C-F bonds are polar because carbon and fluorine differ in their electronegativity, the overall CF4 molecule is non-polar. CH2Cl2 polar molecule The molecule is tetrahedral due to 4 electron repulsions around the central C atom.ĬF4 is a nonpolar molecule. As these polar bonds are symmetrical about the C atom in CCl4, the effect of them is cancelled, so the molecule is non-polar. 
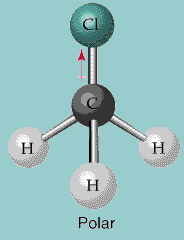
The C–Cl bond is polar, due to the difference in electronegativity of C and Cl. 
The C-Cl covalent bond shows unequal electronegativity because Cl is more electronegative than carbon causing a separation in charges that results in a net dipole. Yes, Methyl chloride (CH3Cl) or Chloromethane is a polar molecule. CH3OH is also used as a gasoline additive in several countries due to its low melting point.Chlorine Monofluoride (ClF) Bond Polarity Electronegativity (F)Ġ.8 Non-Polar Covalent = 0 0 It is used as an anti-freeze in pipes.Methanol is also used for producing hydrocarbons and for the synthesis of other chemicals such as formaldehyde.The smell of this compound is on the sweeter side as compared to ethanol.It is a polar solvent and is also known as wood alcohol because it was once produced by the distillation of wood.The molecular weight of Methanol is 32.04 g/mol.The melting point of Methanol is -97.6 ☌.The boiling point of Methanol ( CH3OH) is 64.7 ☌.Now that we know the polarity of the CH3OH molecule, let us go through some of its physical properties: But as there is a bent in the shape of Methanol, it leads to the formation of an asymmetric structure resulting in the net electric dipole moment’s negative end towards the Oxygen atom. A non-polar molecule has a symmetrical structure, as the dipole-dipole moment is canceled out. This causes a net dipole pointing towards the Oxygen atom, making CH3OH polar.Īpart from the electric charges, the molecule of CH3OH is asymmetrical, which cancels out the possibility of non-polarity. Oxygen has more electron density due to its two lone pairs of electrons. But Oxygen is more electronegative than Carbon or hydrogen in the compound.ĬH3OH cannot be non-polar because there is a difference in electric charges among the atoms in the methanol molecule. Here both Carbon and Oxygen atoms ( which are considered as geometric centers for this compound) in Methanol are electronegative atoms. This net dipole moment can be known by noticing the electric charges on the atoms in the molecule. The polarity of any given compound depends on the net dipole moment on the compound.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
